What is the name of the process when an acid and an alkali react?

When two opposite chemical, acid and alkalis, engages in a process, then this reaction is called Neutralization. When both alkali and acid are mixed, it is like magic happens and it creates something like salt and water. Alkali is known because of its bitter and slippery nature, while acid is known because of its sour and corrosive nature.
Let’s delve into the world of chemistry where you completely know the explanation of What is the name of the process when an acid and an alkali react and reveal all the neutralization complexities.
What is the name of the process when an acid and an alkali react?
When acid and alkali react, Neutralization is the reaction that occurs. Now, we will show you the process of reaction through an equation. Let’s have a look at the reaction
Acid + Alkali Equation:
Acid + Alkali -> Water + Salt
In the following equation, Salt and Water are made when the acid and alkali react. Alkali is the reaction involved in the process and salt is produced because of acid.
Acid and Alkali Reaction Examples:
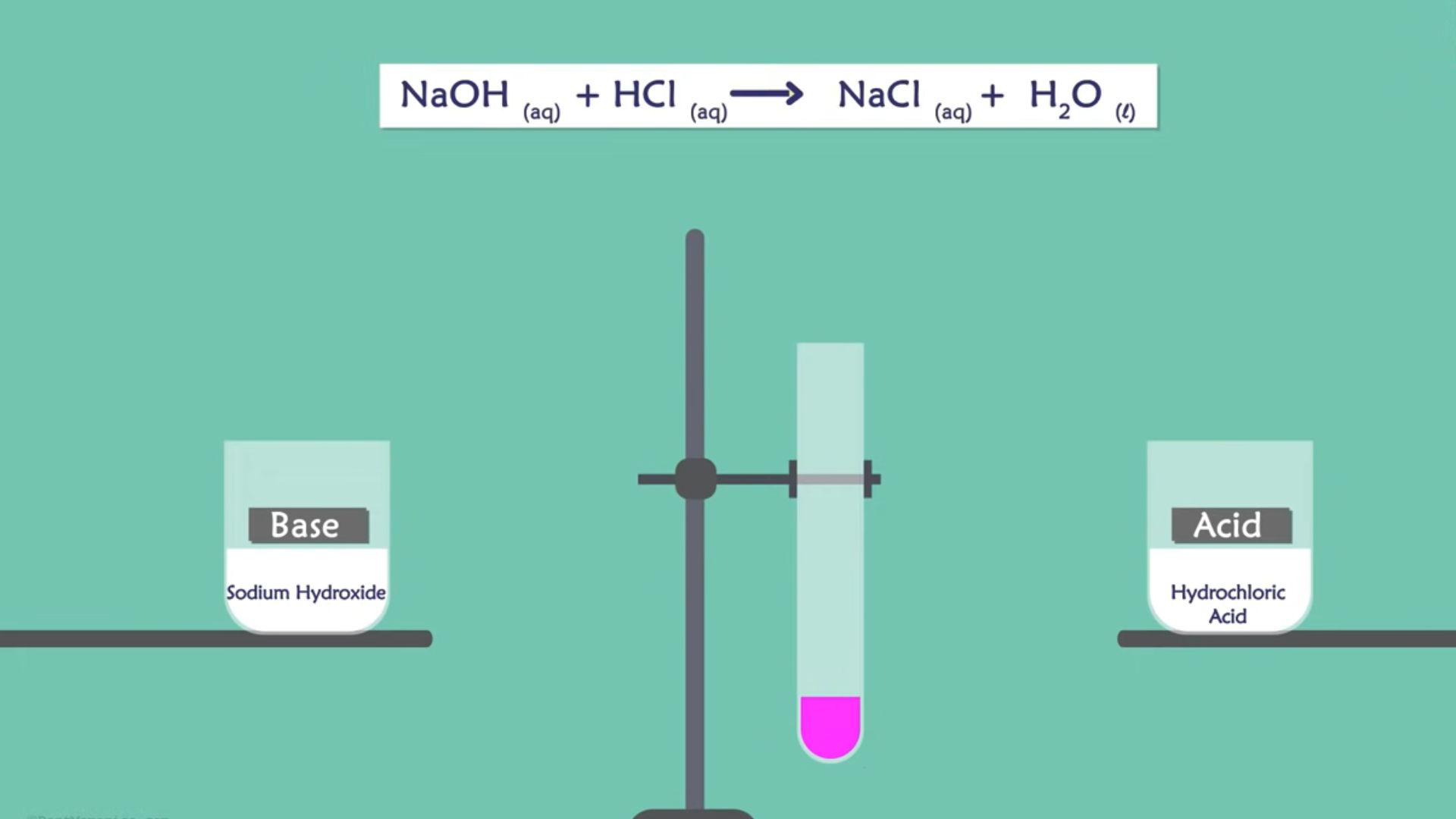
Here, we will show you one of the examples of acid and alkali reactions. Consider the neutralization of HCI (Hydrochloric Acid) with NaOH (Sodium Hydroxide).
HCI + NaOH -> H2O + NaCI
In the above reaction, sodium chloride and water are formed. The Hydrogen ions and acid are combined from the alkali and form water. On the other hand, Chloride and Sodium are combined and form sodium chloride.
What is the Role of pH in this Reaction?
pH is a scale that measures the alkalinity and acidity of a solution. It inherently involves a shift in pH towards the neutrality of the neutralization process. On the other hand, after the neutralization reaction, the pH result of the solution is close to 7. Moreover, it also indicates the balance between alkalinity and acidity.
What is produced when an acid reacts with an alkali?
Salt and Water are produced when you mix acid and alkali. Moreover, the name of the process of mixing the acid and alkali is called neutralization. Now, you surely get your answer about What is the name of the process when an acid and an alkali react.
Frequently Asked Questions:
Q. What is the reaction between an acid and an alkali called?
The reaction between acid and alkali is called Neutralization. You can clearly see the neutralization reaction in the following equation.
H+ + A− + B+ + OH− → AB + H2O
Q. What is the process that takes place when an acid reacts with an alkali?
When the acid and alkali reacts, it forms a Salt and water.
Q. What do acids and alkalis react together to form?
The aklai and acid react together to make salt and water.

Q. What is it called when an acid and base react?
When acid and base react, this process is called the neutralization process.
Final Remarks:
This Reaction is a backbone of many industrial processes. The captivating chemical ballet between alkalis and acids react, Neutralization is the fundamental process that occurs after this reaction. However, the equation of this reaction is;
H+ + OH- -> H2O
This chemical reaction underscores the interconnections of acid and alkali reactions. However, we ensure that you must come to know completely about what is the name of the process when an acid and an alkali react. The significance and elegance of these opposite chemical reactions create a delicate balance.

